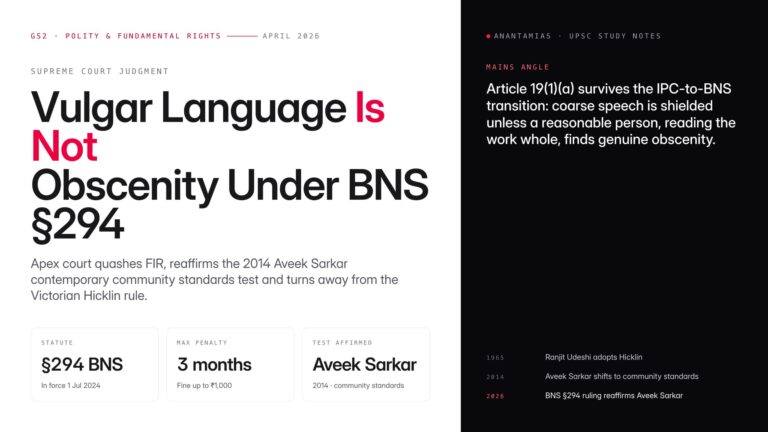
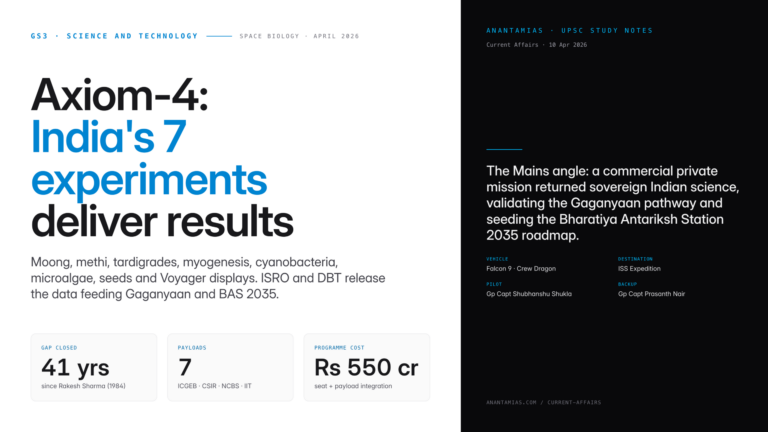
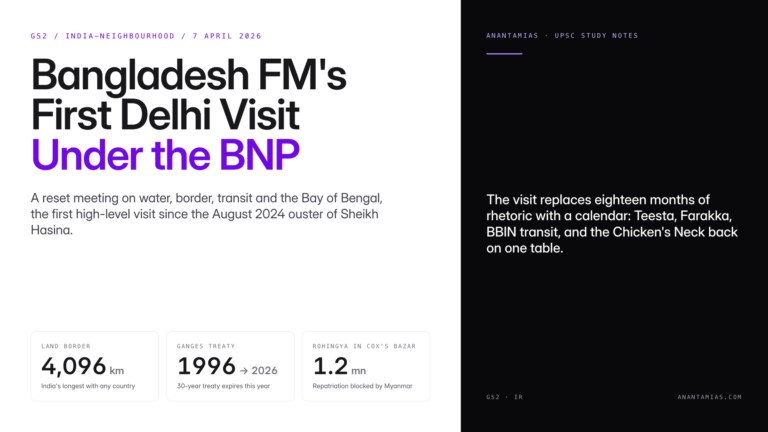
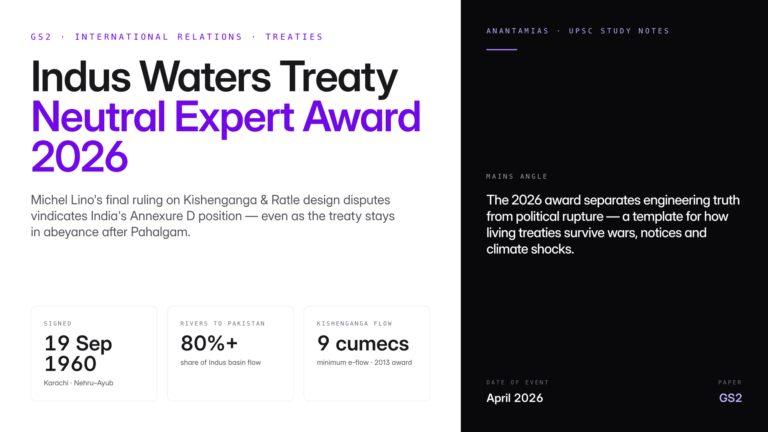
Why in News?
New research has confirmed that genetically modified mosquitoes can suppress malaria parasites from real-world infections. This implies that advanced mosquito genetic engineering can be carried out in malaria-endemic regions for malaria control.
| UPSC Relevance: GS-3 Science and Technology: Biology and Biotechnology Prelims: Malaria; Gene Drive Technology in Malaria Elimination; CRISPR-Cas9 Mains: Gene Drive Technology: Applications & Concerns |
Key facts about Malaria:
- Malaria is a neglected tropical disease, i.e, mainly prevalent among impoverished communities in tropical areas.
- The life-threatening disease is caused by Plasmodium parasites and is transmitted to people by bites of infected female Anopheles mosquitoes.
- Transmission: Through blood transfusion, organ transplant, shared use of needles or syringes contaminated with blood. The malaria parasite is found in red blood cells (RBCs) of an infected person.
- Vaccine: RTS,S/ AS01 (brand name Mosquirix) and R21/Matrix M.
- According to the WHO World Malaria Report 2023, there were approximately 249 million malaria cases and over 6 lakh deaths globally in 2022.
Despite decades of conventional interventions (insecticides, bed nets, and antimalarial drugs), eradication remains elusive, largely due to vector resistance and parasite adaptation.
In this context, Gene Drive Technology has emerged as a potentially transformative tool in Malaria Elimination. It offers the prospect of genetically modifying or suppressing mosquito populations at the ecological level.
What is Gene Drive Technology?
- In standard inheritance, an organism normally has a 50% chance of passing a specific gene to its offspring. Gene drive alters this rule.
- Gene drive is a genetic engineering technique. It is used to alter the law of inheritance to pass on a particular genetic trait from one generation to the next generation, faster than the normal 50/50 inheritance rate.
How does Gene Drive Technology work?
- Gene drives use CRISPR-Cas9 (a molecular scissors tool) to cut the corresponding gene on the chromosome inherited from the other parent.
- The cell’s own repair machinery copies the modified sequence into that gap (through a process called homology-directed repair).
- This conversion typically occurs in the germline (the cells that produce sperm or eggs). This ensures that the drive allele is now present in both chromosomes — and thus passed to nearly 100% of offspring.
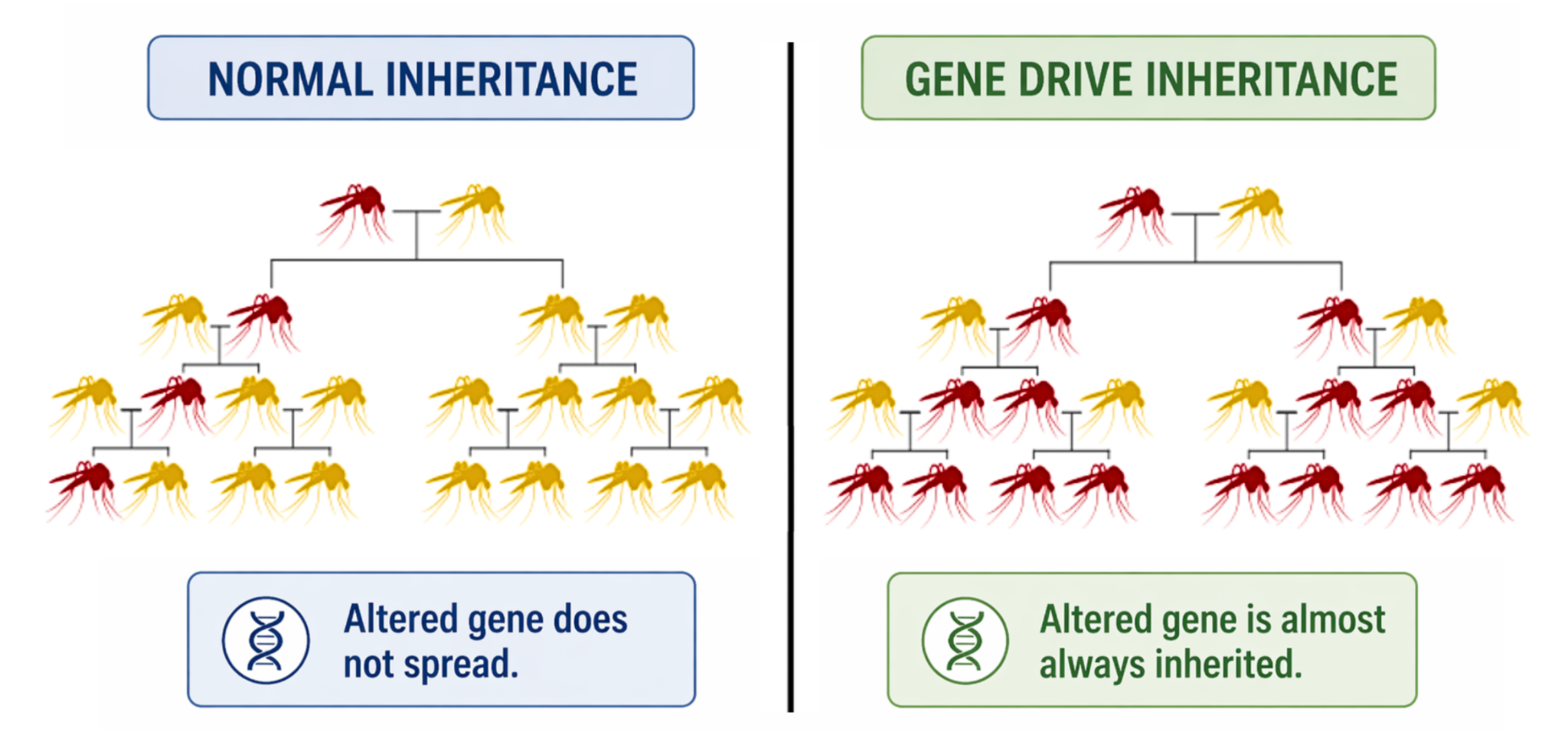
Targeting Malaria: Application of Gene Drive Technology:
- Target Gene: Scientists have targeted a highly conserved gene called doublesex (Agdsx) in Anopheles gambiae mosquitoes.
- Sex-Specific Impact: Disruption of this gene blocks female development but does not affect males. Females with two copies of the modified gene (homozygous) develop an “intersex” phenotype.
- Resulting Traits: These intersex females are sterile; they have male-like mouthparts (making them unable to bite) and do not have functioning ovaries (making them unable to lay eggs).
The mosquito population typically crashed completely by the 8th to 12th generation, as no more fertile females remained to produce offspring.
Types of Gene Drives for Malaria:
Researchers are developing two main types of mosquito gene drives.
- Population suppression: These drives disrupt the genes essential for female mosquitoes to develop or become fertile. As the drive spreads, more females become sterile, causing mosquito populations to shrink or collapse.
- Population modification (also called replacement): Here, mosquitoes remain alive but carry genes that prevent the malaria parasites from developing inside their bodies. This strategy thus reduces the mosquitoes’ ability to transmit malaria, without eliminating the species.
Key research focuses on Anopheles gambiae (primary malaria vector in Africa) and Anopheles stephensi (urban malaria vector in South Asia, including India).
Why is Gene Drive relevant for India?
India accounts for approximately 2% of global malaria cases. India has made significant strides under the National Vector Borne Disease Control Programme (NVBDCP) and the National Framework for Malaria Elimination (NFME) 2016-2030, which targets malaria-free status by 2030.
- Anopheles stephensi invasion: This urban malaria vector, traditionally limited to South Asia, has spread to African cities, making it a global threat. India hosts the native range and must lead research on it.
- Drug and insecticide resistance: Resistance to chloroquine and pyrethroids is documented in Indian states like Odisha, Jharkhand, and Chhattisgarh.
- Tribal and forested belt: Conventional vector control is difficult in high-burden, hard-to-reach tribal areas of central and northeastern India.
- Climate change: Expanding vector habitats into newer geographies (Himalayan foothills, urban areas) demand next-generation tools.
Advantages of Gene Drive Technology:
- Self-propagating: Once released, it spreads without continuous human intervention — unlike insecticide spraying.
- Species-specific: Designed to target specific mosquito species, minimising off-target ecological impacts.
- Cost-effective at scale: One-time release could have generational effects, unlike recurring pesticide campaigns.
- Resistance-breaking: Targets essential genes that mosquitoes cannot easily mutate away from.
- Complements existing tools: Works alongside bed nets, vaccines (e.g., RTS,S/ AS01 – Mosquirix), and antimalarials.
Concerns and Challenges:
Scientific Challenges:
- Off-target effects: CRISPR may cause unintended gene edits in non-target species through horizontal gene transfer.
- Ecological unpredictability: Eliminating a mosquito species could disrupt food webs — birds, bats, and aquatic larvae consumers, etc.
Ethical Concerns:
- Irreversibility: Once released into the wild, population suppression drives may be impossible to reverse.
- Consent of local communities: Indigenous and rural communities affected by the change deserve free, prior, and informed consent (FPIC).
- Cross-border spillover: Mosquitoes do not respect national boundaries — a release in one country could affect another.
- “Playing God” debate: Deliberate extinction of a species raises deep bioethical questions.
Regulatory Gaps:
- No global governance framework: Neither the Convention on Biological Diversity (CBD) nor the Cartagena Protocol on Biosafety provides specific guidance on gene drives.
- India’s regulatory vacuum: India’s current Environment Protection Act, 1986, and Rules for the Manufacture, Use, Import, Export and Storage of Hazardous Microorganisms (1989) do not explicitly cover gene drives.
- Dual-use concerns: Gene drive technology could theoretically be weaponised (bioweapon risk).
Way Forward:
- Develop a national regulatory framework: India must establish clear biosafety norms for gene drive research, ideally under a dedicated biotechnology governance authority or an expanded GEAC (Genetic Engineering Appraisal Committee) mandate.
- Invest in indigenous R&D: ICMR, DBT, and academic institutions should build capacity in CRISPR-based vector biology, particularly for Anopheles stephensi.
- Community engagement: Any field trial must be preceded by robust community consent mechanisms and transparent risk communication, especially in tribal regions.
- International collaboration: India should engage with the WHO VCAG, Target Malaria, and Global Health Security Agenda (GHSA) for knowledge-sharing and co-governance.
- Biodiversity safeguards: Ecological impact assessments must precede any release, in consonance with India’s obligations under the Biological Diversity Act, 2002.
- Parallel investment in conventional tools: Gene drives are a long-term tool; India must simultaneously strengthen ASHA networks, surveillance systems, and the malaria vaccine rollout.
As India strives toward its 2030 malaria elimination goal, gene drives could be a powerful arrow in the quiver, but only if wielded with scientific rigour and ethical responsibility.
UPSC PYQ 2023
Q. ‘Wolbachia method’ is sometimes talked about with reference to which one of the following?
(a) Controlling the viral diseases spread by mosquitoes
(b) Converting crop residues into packing material
(c) Producing biodegradable plastics
(d) Producing biochar from thermo-chemical conversion of biomass
Answer: (a)